Product Deveopment Pocket Guide
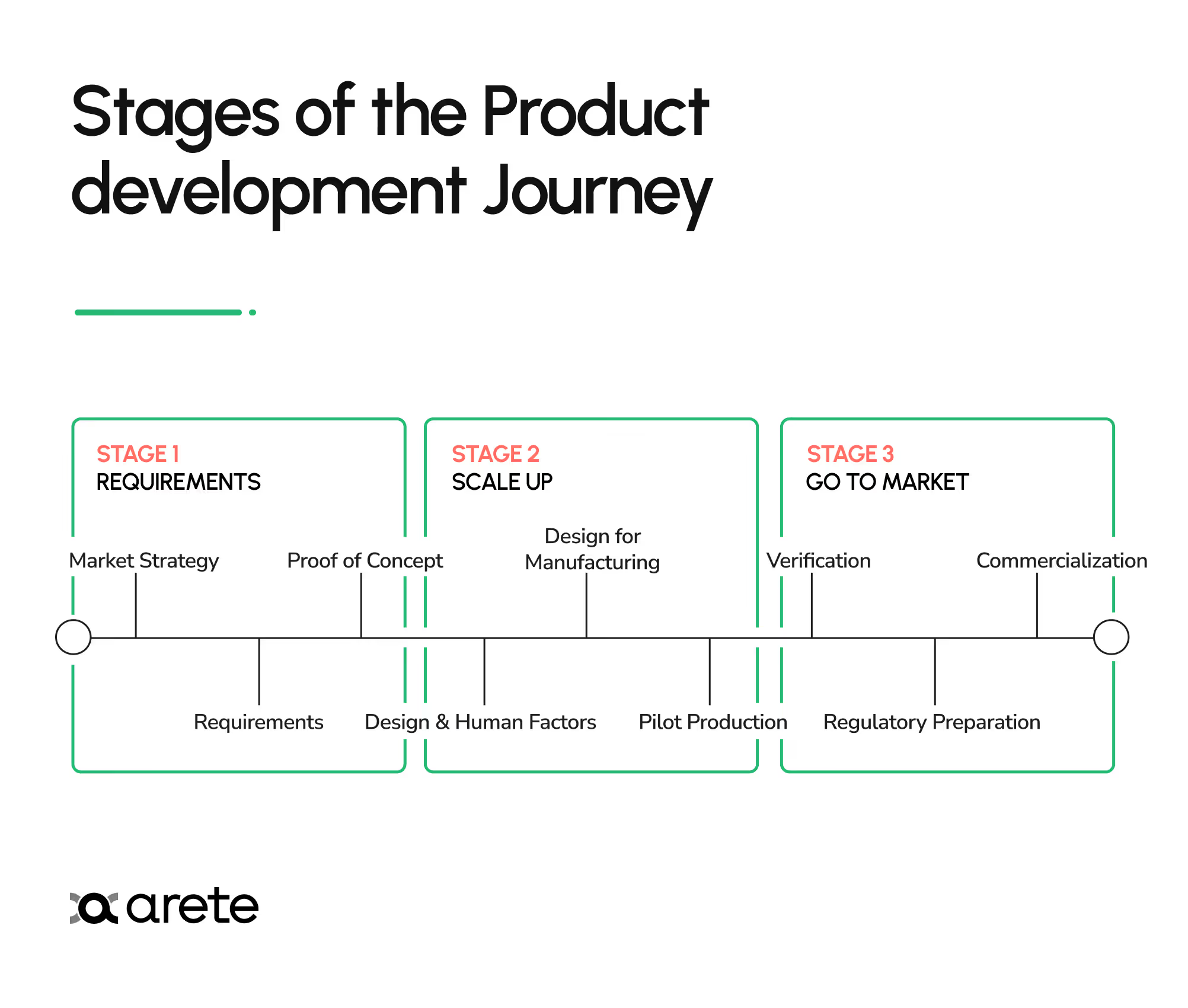
The Three Stages of Product Development
Download the Full Pocket Guide PDF
Stage 1: Requirements
Stage 2: Scale Up
Stage 3: Go to Market
What matters is maintaining discipline throughout: documenting decisions, managing risk continuously, and keeping cross-functional teams aligned. The companies that navigate this journey most effectively aren't necessarily the ones with the biggest budgets. They're the ones that plan thoughtfully, collaborate well, and learn quickly from setbacks.
FAQs About Product Development
What are the main stages of product development?
Product development typically moves through three stages: Requirements (defining the problem, specifications, and proving feasibility), Scale Up (integrating components, refining design for manufacturing, and running pilot production), and Go to Market (verification testing, regulatory preparation, and commercialization). Each stage builds on the previous one, so thoroughness early prevents costly rework later.
How long does the product development process take?
Timelines vary significantly based on device complexity, regulatory classification, and technical challenges. Simple Class I devices might reach market in 12–18 months, while complex Class II or III devices often require 3–5 years. Strong planning, early risk identification, and integrated cross-functional teams can help compress timelines without cutting corners.
When should I involve regulatory and quality teams?
As early as possible. Teams that treat regulatory strategy as a late-stage activity often face preventable delays. Engaging QA/RA during requirements definition helps ensure design decisions align with submission expectations, documentation stays current, and risk management remains integrated throughout development.
What's the difference between verification and validation?
Verification confirms you built the product correctly — testing whether the device meets its design specifications. Validation confirms you built the right product — demonstrating that it meets user needs in real-world conditions. Both are essential for regulatory submissions and should involve representative users and samples.
Why do so many product development projects get delayed?
Common culprits include poorly defined requirements, underestimating regulatory complexity, late-stage design changes, supply chain issues, and inadequate documentation. Most delays trace back to insufficient work in early stages — rushed feasibility assessments or unclear specifications that create compounding problems downstream.
Should we build internal capabilities or partner with a development firm?
It depends on your team's expertise, timeline, and risk tolerance. Many startups and emerging companies partner with experienced development firms to access multidisciplinary capabilities — engineering, regulatory, quality, manufacturing — without building those functions internally. This can accelerate timelines and reduce risk, especially for teams navigating regulated development for the first time.
Partner With Arete Biosciences




